The Standards for Cosmetics Efficacy Claims Evaluation (hereinafter referred to as the "Standards") have divided cosmetics efficacy claims into four classes based on the degree of their efficacy. The stronger the efficacy claimed, the more adequate the evidences needed to be provided.
On April 9, the National Medical Products Administration released three important announcements: the Cosmetics Classification Rules and Catalogue, the Standards for Cosmetics Efficacy Claims Evaluation and the Technical Guidelines of Cosmetics Safety Evaluation (2021).As supporting bills for the newly promulgated Regulations on Supervision and Administration of Cosmetics, these three announcements define cosmetics classification rules and technical standards for evaluation of efficacy claims and safety in a detailed and specific way, and they are about to take effect officially on May 1.
01
Subdivision of cosmetics:
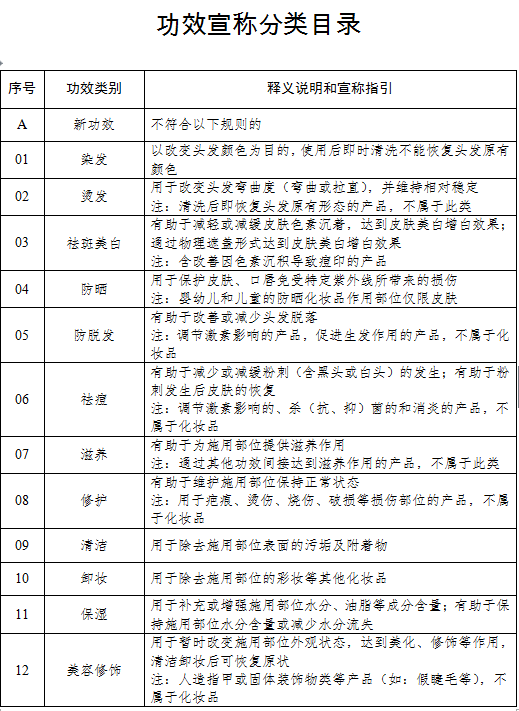
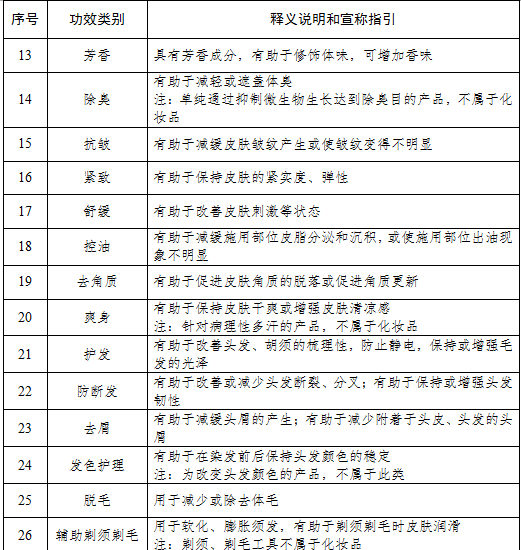
Involving 26 efficacy types and 11 product formulationsThe Cosmetics Classification Rules and Catalogue (hereinafter referred to as the "Catalogue") has broken down the cosmetics on the basis of efficacy claim, site of action, targeted user, product formulation and method of application, and different codes have been given to different efficacy types.The cosmetics to be registered/filed will be given corresponding serial numbers based on the above-mentioned levels, such as efficacy and site of action. Different groups of catalogue code can be connected with "-" to form a complete product classification code.In terms of efficacy claims, the Catalogue has divided cosmetics efficacy into 26 types: hair dye, perm, whitening, sun protection, hair loss prevention, acne treatment, nourishing, repair, cleaning, makeup removal, moisturizing, retouching, fragrance, deodorization, wrinkle prevention, firming, soothing, oil control, exfoliating, body powder, hair care, hair breakage prevention, dandruff prevention, hair color care, hair removal and aided shaving & hair removal.Compared with the efficacy types that appeared in the previous version of the Catalogue, the type of "hair style" is removed; two types of "whitening" and "whitening (only physical mask)" are combined into one single type to refer to the effect of reducing or slowing down skin pigmentation in order to whiten the skin, or to do so via physical mask (including products for improving acne marks caused by pigmentation).In terms of product formulations, freeze-dried formulations such as freeze-dried powder and slices have also been included in the 11 specified formulations. The main cosmetics product formulations include paste/cream/lotion, liquid, gel, powder, lump, mud, ceryl, spray, aerosol, patch and film (including base material).Moreover, as to the classification of targeted users, the Catalogue has divided user population into infants (0 – 3 years old, including 3 years olds), children (3 -12 years old, including 12 years olds) and ordinary people. Different rules have also been introduced in cosmetic efficacy claims for users from the infant and children groups. For cosmetics for infants, efficacy claims can only be limited to cleaning, moisturizing, hair care, sun protection, soothing and body powder; for cosmetics for children, efficacy claims can only be limited to cleaning, makeup removal, moisturizing, retouching, fragrance, hair care, sun protection, repair, soothing and body powder. Additionally, cosmetics claimed to be suitable for use by pregnant and breastfeeding women should be registered and administered as special cosmetics.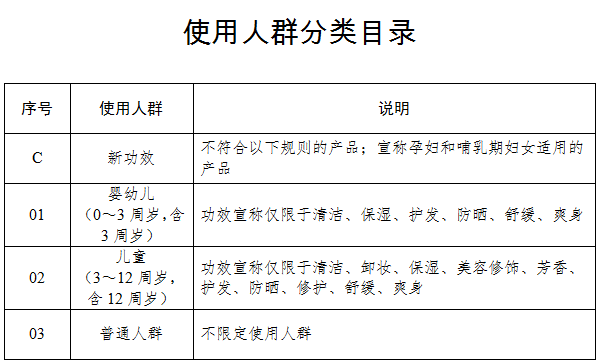
It should be noted that when a product has more than one type of efficacy claim, site of action, target user or formulation, it can use multiple S/Ns which should be arranged in proper order and connected with "/".The Catalogue also indicates that the registrants of the cosmetics registered or filed before May 1, 2021 should submit product classification codes on the cosmetics registration and filing information service platform before May 1, 2022.
02
Efficacy claims should be supported with scientific evidence based on classesAccording to the Standards efficacy claims of cosmetics should be supported with adequate scientific evidence, mainly including literature documentation, research data or cosmetic efficacy claim evaluation test (human body efficacy evaluation test, consumer use test and lab test) results.Registrants can perform cosmetics efficacy claim evaluations on their own or by commissioning competent evaluation bodies based on requirements for different cosmetics efficacy claim evaluation items and upload the abstracts of the evidences of their product efficacy claims to the special website designated by the National Medical Products Administration as required.The Standards have divided cosmetics efficacy claims into four classes based on the degree of their efficacy. The stronger the efficacy claimed, the more adequate the evidences needed to be provided.Class 1 cosmetics feature efficacy claims that can be identified directly via vision and smell (such as cleaning, makeup removal, retouching, fragrance, body powder, hair dye, perm, hair color care, hair removal, deodorization and aided shaving & hair removal), or that take effect via simple physical masking, attaching or friction (such as physical mask for whitening, physical exfoliating and physical pore cleansing) with clear indications on their labels to have only physical effect. These cosmetics may be exempted from releasing abstracts of evidence for product efficacy claims.Class 2 cosmetics only have moisturizing and hair care efficacy and their efficacy claim evaluations can be performed via literature research, data research & analysis or cosmetics efficacy claim evaluation tests.Class 3 cosmetics are those with wrinkle prevention, firming, soothing, oil control, exfoliating, hair breakage prevention and dandruff prevention functions and claimed to be mild and not irritating or with quantified indexes, such as retention time of efficacy claims. Their efficacy claims should be evaluated via cosmetics efficacy claim evaluation tests alone or in combination with literature or research data analysis results.Class 4 cosmetics are those with whitening, sun protection, hair loss prevention, acne treatment, nourishing and repair functions and with specific claims (such as claimed to be suitable for sensitive skin or use tear-free formula). Their efficacy claim evaluation can be carried out via human body efficacy evaluation tests. For cosmetics with specific claims, consumer use tests can also be used for efficacy claim evaluation.Furthermore, for cosmetics with new efficacy, a cosmetics registration and filing inspection body should be engaged to perform human body efficacy evaluation tests according to the requirements in the compulsory national standards and technical standards. Meanwhile, test reports should also be produced.If test methods other than those specified in the compulsory national standards and technical standards are used for testing products with new efficacy, the registrants in question should commission two or more cosmetics registration and filing inspection bodies to validate such methods. Only those validated to be conforming can be used to evaluate new product efficacy and such parameters as effectiveness and reliability of such methods should be described in the product efficacy claim evaluation reports produced.It is worth noting that a period of transition has been provided by theStandards.Starting from January 1, 2022 the cosmetics registrants applying for special cosmetics registration or filing for ordinary cosmetics should perform efficacy claim evaluations on their products as per the requirements in the Standards and upload the abstracts of the evidences of their product efficacy claims to the special website designated by the National Medical Products Administration as required.The registrants of cosmetics registered or filed before May 1, 2021 should perform efficacy claim evaluations on their products as per the requirements in the Standards and upload the abstracts of the evidences of their product efficacy claims before May 1, 2023.The registrants of cosmetics registered or filed between May 1, 2021 and December 31, 2021 should perform efficacy claim evaluations on their products as per the requirements in the Standards and upload the abstracts of the evidences of their product efficacy claims before May 1, 2022.
03
Multi-color makeup products
can share their efficacy claim evaluation test data.For efficacy evaluation of multi-color makeup products, an area of concern of the industry, the Standards have officially provided a solution.The Standards specify that, multi-color series makeup products from the same registrant (with the exception of products claimed to have acne treatment, nourishing and repair functions) can share efficacy claim evaluation test result data as the evidences of their efficacy claim evaluations when equivalent evaluation conditions and requirements are satisfied.Equivalent evaluation applies to multi-color makeup products from the same registrant sharing same kind and content of basic formula as well as series name, but with different types or content of colorant (including color tone change).For such makeup products the efficacy claim evaluation test can be performed in the form of spot check, with the sampled number of colors for check no less than 20% of the total number of colors in the series (when the total color number is less than 5, it should be considered as 5 colors). The products with the minimum content of colorant should be selected with preference for the efficacy evaluation test and a test report should be produced. For products applicable for equivalent evaluation, an equivalent evaluation report and an efficacy claim evaluation test report should be retained for future inspection."This, to some extent, helps cut the efficacy evaluation test cost of makeup enterprises and ensures product safety in a scientific manner," an industrial insider pointed out. "For enterprises with a history of attaching importance to efficacy evaluation, it gives them greater competitive edge; while for those neglecting such evaluation, they will have a hard time adapting to stricter market supervision."Jiang Ligang, CTO of PROYA Cosmetics Co., Ltd. once predicted that in the future emphasis would be based on science and evidence and product efficacy claims would be consistent with their actual functions. "Validated via evaluation" would become a major marketing expression to attract consumers. Cosmetics would also become more like pharmaceuticals, with single and well-defined functions; cost of developing new products would increase significantly. New product launching and updating would slow down, boastful innovations would decrease and more attention would be paid to the actual demands of consumers.A safer, more segmented and practical cosmetics market is shaping up.